 The Whitney Laboratory for Marine Bioscience
The Whitney Laboratory for Marine Bioscience

Paul Linser graduated with a B.S. in biology from the University of Cincinnati. His graduate work was also done at the University of Cincinnati, where he earned his Ph.D. in developmental biology. Linser’s postdoctoral research was done with Aaron Moscona at the University of Chicago.
Email: pjl@whitney.ufl.edu
Mosquitoes carry malaria, West Nile virus, Dengue fever and a variety of other serious diseases. Forty-one percent of the world’s population lives in areas where malaria is transmitted and 350-500 million cases of malaria occur worldwide each year. In Africa alone, each year, malaria is responsible for over 2,700 deaths per day, or 2 deaths per minute.
Part of the strategy to eliminate or reduce the incidence of these diseases is to develop environmentally safe treatments that will target mosquitoes selectively, without harming humans, livestock and other insects, particularly agriculturally important species such as bees. For this purpose, one very attractive feature of mosquitoes is that their larvae rely on alkaline digestive fluids to break down food in their digestive systems. However, almost all other animals use acidic digestive fluids.
What if… we were able to develop new generations of insecticides that are able to target the alkaline conditions of the mosquito larvae digestive system? These new insecticides would kill mosquito larvae but leave other insects, fish and humans unaffected.
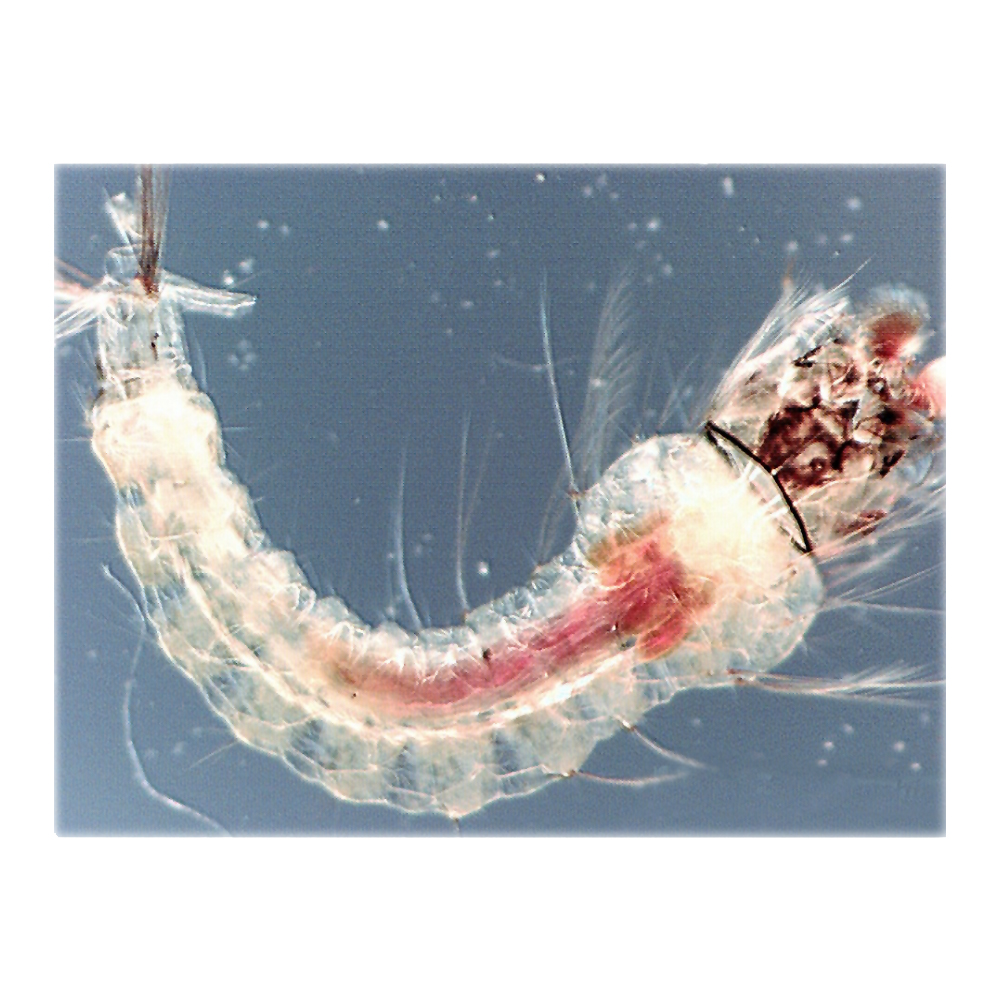
The mosquito larva is transparent, allowing us to visualize the entire digestive system in the whole animal. The pink area indicates the alkaline section of the gut where digestion takes place.
As part of this effort to control mosquitoes, my lab is trying to understand how the mosquito larva is able to make its digestive system so extremely alkaline, and to use this information to identify mosquito-specific molecules that could be used in new generations of mosquitocides to selectively kill mosquito larvae. Think of what this would mean for the control of malaria and the many other diseases carried by this most dangerous of insects.
The Whitney Laboratory is situated on a narrow barrier island on the northeast coast of Florida, adjacent to Marineland. The island habitat is replete with many aquatic organisms in the adjacent oceanic and intracoastal/estuarine environments. Among the local “swimmers” are species of larval insects that use the salt marsh and its tributaries as breeding grounds. Mosquito larvae are prominent in the shallows of the estuary and its many creeks and fresh water feeders. Mosquitoes have been a concern for humanity for our entire existence, producing a tremendous nuisance and impediment to comfortable living. Beyond the nuisance and comfort perspective, which is quite important in coastal Florida environments, mosquitoes are the number one threat to human health worldwide according to the World Health Organization (WHO). Mosquitoes vector (i.e. transmit) numerous viral and parasitic diseases and are responsible for millions of deaths per year. Malaria alone kills one to two million people per year worldwide. This laboratory works on the fundamental cell and molecular biology of mosquitoes with an emphasis on the aquatic life phase, that is the larvae.
Mosquito larvae, as aquatic organisms, live in a varied and often hostile environment. The mechanism for acquiring nutrients for growth and metamorphosis involves many details. We work specifically on the digestive strategy of mosquito larvae. Larval insects are at the simplest level stomachs with an exoskeleton as their fundamental purpose is to eat enough to fuel the development of a reproductive adult. Mosquito larva have numerous specific digestive attributes that present interesting scientific questions as well as potential targets for novel control strategies. Our laboratory focuses on the relatively novel quality of larval digestion that is based on an internal pH of the anterior half of the stomach that is extremely basic (pH around 10.5). This is one of the highest pH extremes found in biology. Understanding how the larval gut generates, maintains and functions with this high luminal pH is part of our research direction.
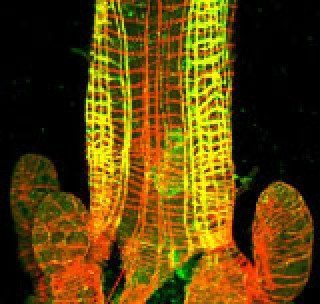
The gut of a mosquito larva fluorescently labeled to show the muscles. Yellow = the presence of carbonic anhydrase, red = absence of CA.
We utilize the tools of molecular biology, cell and tissue physiology and advanced imaging technology to identify and characterize major gene products in the larval mosquito gut and their specific functions. From comprehensive transcriptomic analyses to cloning specific messenger RNAs to producing immunological probes for the proteins coded by those genes, we strive to build a holistic picture of how specific gene products and their tissue and cellular localization contribute to the functions of the gut and other interactive tissues and cells. Among the key players in the alkaline gut of mosquito larvae is a family of genes who produce enzymes of the carbonic anhydrase family. These enzymes are intimately involved in pH regulation as they interconvert carbon dioxide (the major end product of metabolism) and ions such as bicarbonate and carbonate. It is the carbonate anion aligned with a strong cation such as potassium that actually buffers the pH at levels as high as 10.5.
To date we have characterized 8 of the 12 carbonic anhydrases encoded in the mosquito (Anopheles gambiae) genome. Comprehensive gene expression profiling (i.e. DNA microarray analyses) has also been generated which provides the “big picture” of gene expression in relation to homeostasis in the various cell types of the larval gut. New emphases that have emerged from the big picture analyses included a focus on gut function in mediating innate immunity, the role of the salivary glands in gut function and homeostatic ion balance as a function of the activity of the hindgut (rectum) of larvae. In addition, we are performing similar analyses in adult mosquitoes in an effort to understand ionic homeostasis following blood feeding and the steps toward egg production.
For the first 25 years at the Whitney Lab, the Linser group investigated specific developmental questions in the vertebrate nervous system. This project overlapped with the mosquito emphases for a few years but was inactive for several of the last 5 years. In collaboration with Dr. Shinichi Someya of the University of Florida Institute on Aging, we are again studying developmental questions in the vertebrate nervous system. Specifically, we are investigating the degenerative changes that occur in the inner ear with advanced age. This represents a new and very exciting direction that takes advantage of our past studies in the nervous system during early development. A newly funded NIH project takes us into the realm of development at the other extreme…old age. We are now asking…”What if we could prevent the age-related loss of specific cells in the inner ear and the Organ of Corti”?