 The Whitney Laboratory for Marine Bioscience
The Whitney Laboratory for Marine Bioscience
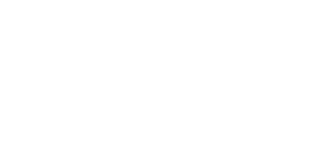

Dr. Martindale obtained a B.A. in natural sciences at New College of the University of South Florida and earned his Ph.D. in zoology at the University of Texas at Austin. After spending 9 years on the faculty at the University of Chicago, Dr. Martindale was recruited the University of Hawaii and ultimately assumed the Directorship of the Kewalo Marine Lab in Kaka’ako. In 2010 Dr. Martindale and Dr. Max Telford initiated an open access journal called EvoDevo (http://www.evodevojournal.com/). Dr. Martindale was named the Director of the Whitney Lab for Marine Bioscience in December 2012.
Allen Distinguished Investigator
Email: mqmartin@whitney.ufl.edu

We are interested in a broad range of problems associated with the cellular, molecular, and evolutionary basis of biological pattern formation. My lab utilizes a variety of molecular and “classical” techniques of microinjection, cell labeling, ablation, and transplantation, to address fundamental problems in developmental biology in a broad phylogenetic context. One area of interest is to understand the evolution of biological novelties and the increase in biological “complexity”. These include the molecular origins of presumably rare evolutionary events such as the formation of the “middle” germ layer (mesoderm), the evolution of elements of the nervous system in the Metazoa, and the evolution of unique cell types (cnidocytes, colloblasts, sensory cells, etc.). Many of these studies utilize cnidarians (the starlet sea anemone Nematostella vectensis) and ctenophores (the lobate Mnemiopsis leidyi), both of whose genomes have been sequenced. We continue to develop functional techniques in these systems as well as identify new models to uncover conserved and novel molecular mechanisms underlying cell type diversification.
We are interested in the degree of spatial information present in unfertilized and fertilized embryos and to what extent the cleavage program is responsible for the segregation of developmental potential. It is clear that different embryos make these decisions at different times in development and that these details are involved in the evolution of new body plans, cell types, and tissue interactions.
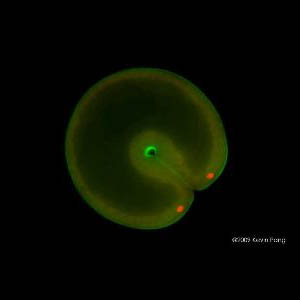
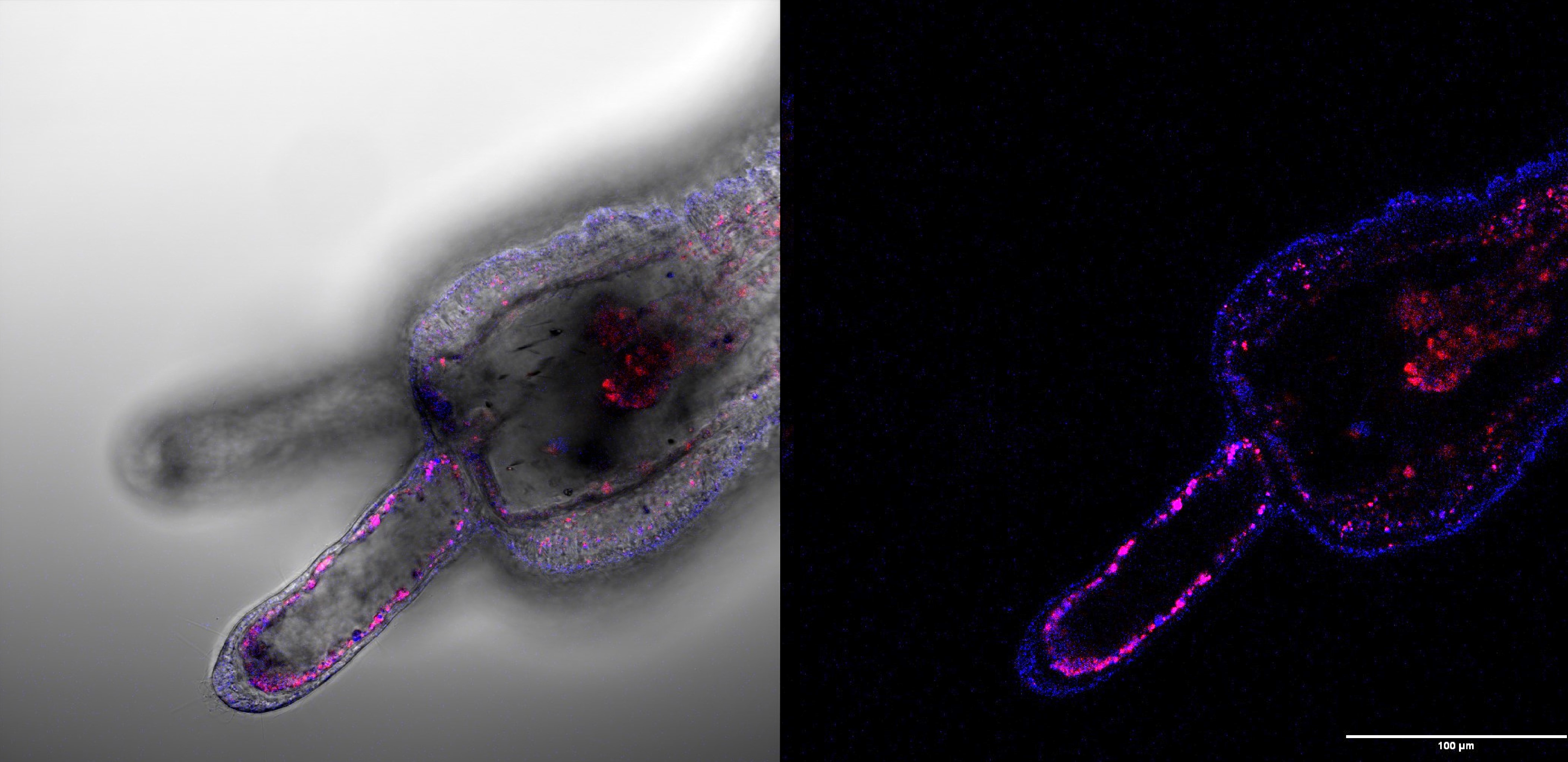
Another area of focus is to understand the relationship between development and regeneration in a variety of marine invertebrates. Does regeneration merely recapitulate the product of ontogeny? Is cell division necessary? Do cells have “memory”? Are there only a small subset of “stem” cells generated during development that can direct regenerative events? Is regeneration an ancient property of metazoans? Or has been “invented” many times in different animal lineages? There are many species whose embryos/larvae cannot replace missing parts, but their adult forms can. How is the ability to regenerate turned on in these adult animals? Most marine metazoans have a high capacity to regenerate and this biological diversity thus provides powerful new opportunities for understanding regenerative biology.

Coral reef bleaching is one of the most drastic and visibly and obvious biological responses to global climate change. Coral bleaching is the breakdown in a symbiotic association of the coral host and a dinoflagellate/algal cell from the genus Symbiodinium. Corals are difficult to work with in the lab and real time cellular level observations are virtually impossible. We have developed a new laboratory model to study coral bleaching in another cnidarian species, the Upside-Down Jellyfish, Cassiopea xamachana.
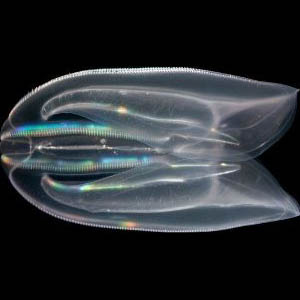
Living organisms can synthesize biominerals in exquisite shapes and a wide range of sizes. Examples come from single celled organisms like diatoms and coccolithophores, snail shells, sponge spicules, vertebrate bones and teeth, and of course coral reefs. These elaborate and precisely shaped structures cannot be readily generated in the lab, even with high temperatures and organic solvents. Surprisingly little is known about the “rules” of biomineralization. The fact that biomineralization has evolved on Earth over 50 times indicates that different organisms have discovered their own ways to generate biominerals. We are a team of chemists, physicists, and soft matter engineers working to understand the cellular and molecular aspects of biomineralization using the favorable properties of a marine invertebrate, the starlet sea anemone Nematostella vectensis. Nematostella is a non-biomineralizing an anthozoan cnidarian relatively closely related to scleractinian corals, and we hope to engineer the biomineralizing process to generate shapes, and even synthetic compounds unknown to Nature as a proof of principle of our understanding of the biomineralization process. Biomineralization is a fascinating subject of interest to young and old students alike and progress on this project will be incorporated into Whitney’s educational activities throughout North Florida.